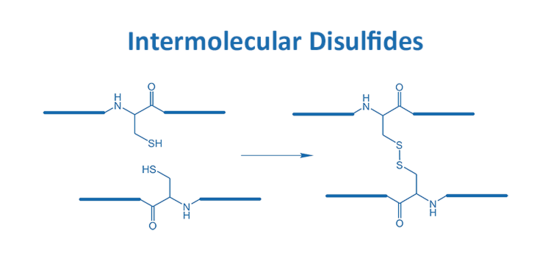
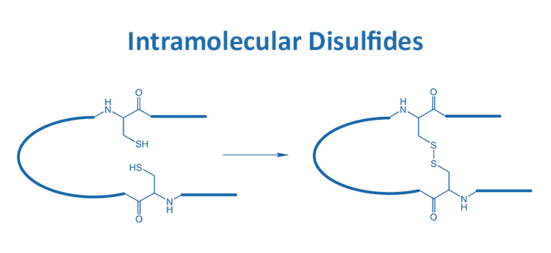
A correct fold is essential for many peptides and is often realized by the implementation of disulfide bonds. Such disulfide bridges appear either inter-molecular or intra-molecularly.
Whereas a single disulfide bond is formed easily, multiple disulfide bonds are a serious challenge. Generally, two strategies exist for forming disulfide bonds. Non-directed oxidation might be carried out in case of larger peptides with a natural fold. Thereby the disulfides are formed statistically, and usually the most thermodynamically stable fold will be the main product.
However, in case of shorter peptides, a stepwise formation of the disulfide pattern is desired. A selective deprotection of cysteines allows a controlled cyclization of the disulfide bonds.
BIOSYNTAN offers peptides with up to 3 disulfide bonds in a defined connection pattern.